Description
Product Information
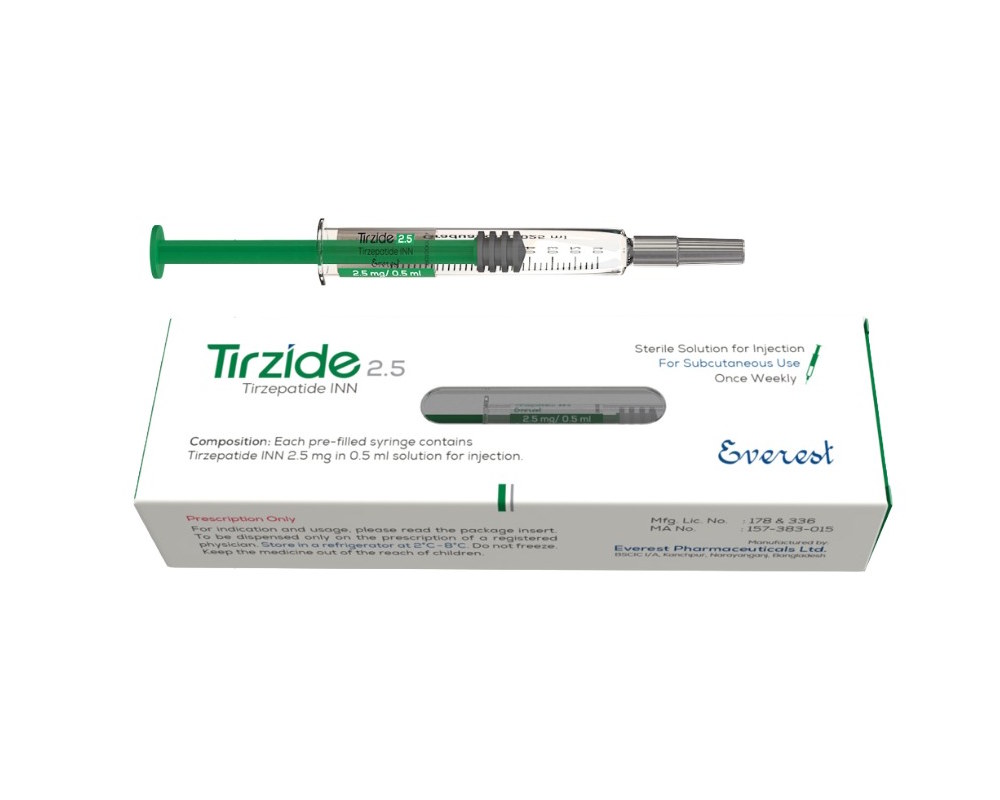
- Product Overview: Tirzide 2.5® is a dual incretin receptor agonist formulation containing Tirzepatide, designed to improve glycemic control in adults with type 2 diabetes mellitus and support weight management under medical supervision.
- Composition: Tirzepatide INN 2.5 mg / 0.5 mL | Dosage Form: Injection
- Pharmacological Class: Dual GIP and GLP-1 Receptor Agonist
- Mechanism of Action: Tirzepatide activates both glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors, enhancing insulin secretion, reducing glucagon levels, slowing gastric emptying, and decreasing appetite.
- Indications: Type 2 diabetes mellitus | Adjunct to diet and exercise for glycemic control | Weight management support in eligible patients
- Dosage & Administration: As directed by physician only | Subcutaneous injection | Dose titration required based on clinical response
- Clinical Benefits: Improves blood glucose control | Supports significant weight reduction | Reduces appetite | Improves metabolic health markers
- Contraindications: Hypersensitivity to Tirzepatide or any component of the formulation | Not recommended in patients with personal or family history of medullary thyroid carcinoma | MEN2 syndrome
- Precautions: Risk of pancreatitis | Monitor renal function in susceptible patients | Not indicated for type 1 diabetes | Use only under strict medical supervision
- Drug Interactions: May delay absorption of oral medications due to slowed gastric emptying | Caution when used with insulin or sulfonylureas (risk of hypoglycemia)
- Special Populations: Not recommended during pregnancy or lactation unless clearly advised by physician | Use in elderly under medical supervision
- Adverse Effects: Nausea | Vomiting | Diarrhea | Decreased appetite | Abdominal discomfort | Rare risk of pancreatitis
- Storage Conditions: Store in refrigerator (2°C to 8°C) | Do not freeze | Protect from light | Keep out of reach of children
- Packaging: Prefilled injection pen / vial (as available)
- Manufacturer: Everest Pharmaceutical Ltd.



Reviews
There are no reviews yet.