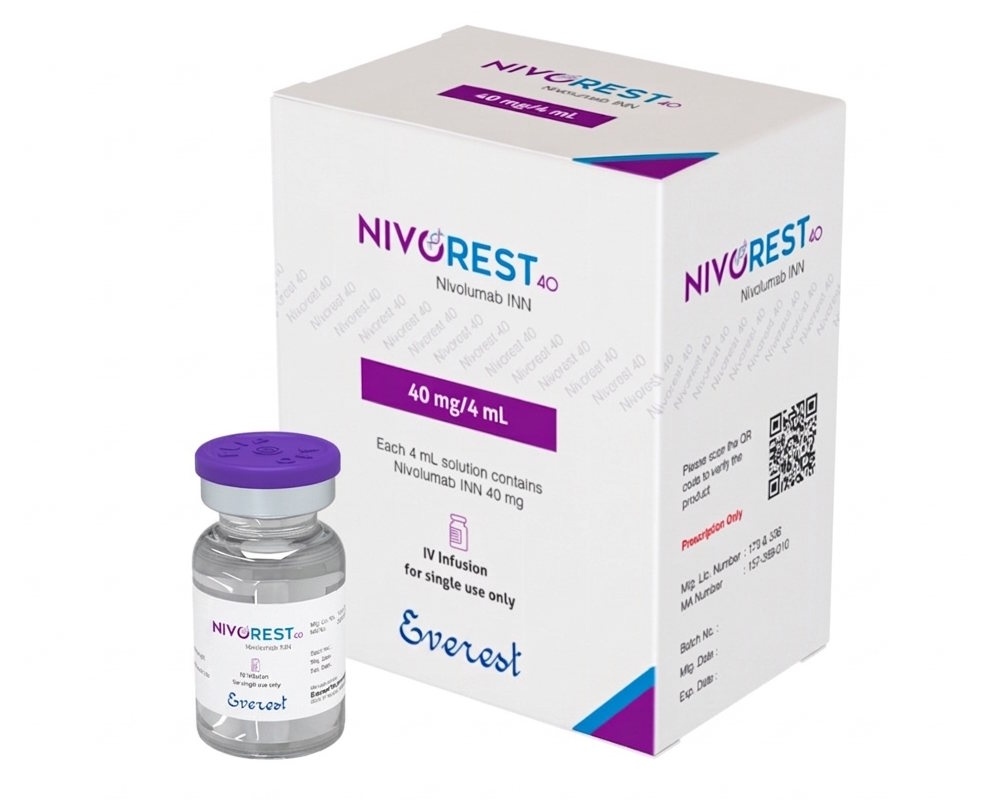
Description
1. Product Overview:
Nivorest® is an injectable formulation of Nivolumab, a monoclonal antibody used in immuno-oncology for the treatment of multiple advanced malignancies, including non-small cell lung cancer, melanoma, renal cell carcinoma, and others.
2. Composition:
Active Ingredient: Nivolumab | Dosage Form: Injection (Concentrate for Infusion)
3. Pharmacological Class:
Programmed Death-1 (PD-1) Immune Checkpoint Inhibitor
4. Mechanism of Action:
Nivolumab blocks the PD-1 receptor on T-cells, preventing cancer cells from evading immune detection. This enhances T-cell mediated immune response against tumor cells, leading to cancer cell destruction.
5. Indications:
Non-small cell lung cancer (NSCLC) | Melanoma | Renal cell carcinoma | Hodgkin lymphoma | Head and neck cancer | Other advanced malignancies
6. Dosage & Administration:
As prescribed by oncologist | Intravenous infusion only | Administered in hospital/clinical setting | Dose and schedule depend on cancer type and patient condition
7. Clinical Benefits:
Enhances immune system response against cancer | Improves survival in advanced cancers | Effective in multiple tumor types | Provides durable response in selected patients
8. Contraindications:
Hypersensitivity to Nivolumab or any component
9. Precautions:
Risk of immune-related adverse reactions (lung, liver, colon, endocrine disorders) | Requires regular monitoring of organ function | Use under strict oncology supervision
10. Drug Interactions:
May interact with immunosuppressants such as corticosteroids, potentially reducing efficacy
11. Special Populations:
Pregnancy: Not recommended | Lactation: Not recommended | Pediatric: Use only under specialist guidance | Elderly: Requires careful monitoring
12. Adverse Effects:
Fatigue | Skin rash | Diarrhea | Immune-related pneumonitis | Hepatitis | Endocrine disorders (thyroid dysfunction)
13. Storage Conditions:
Store at 2°C–8°C (refrigerated) | Do not freeze | Protect from light | Keep out of reach of children
14. Packaging:
Injection vial
15. Manufacturer:
Everest Pharmaceutical Ltd.


Reviews
There are no reviews yet.